
Finding the right information about Diethylaluminum Chloride (DEAC) can be challenging, leading to confusion and ineffective applications. Misunderstandings can result in costly mistakes. We provide clarity on DEAC's versatile industrial uses.
Diethylaluminum Chloride (DEAC) [CAS: 96-10-6] is widely used as a co-catalyst in polymerization processes, pharmaceutical synthesis, specialty chemical manufacturing, and electronics production due to its high reactivity and ability to enhance catalytic efficiency.
Understanding the multiple applications of DEAC can significantly optimize your processes, ensuring efficiency and regulatory compliance. Explore further to grasp its full potential.
What Is Diethylaluminum Chloride and Why Is It Important in Industrial Chemistry?
Diethylaluminum Chloride (DEAC) is often misunderstood or underutilized, creating inefficiencies in industrial chemistry processes. Without clear knowledge, chemical manufacturers risk operational disruptions and safety hazards. Proper understanding can prevent such issues.
Diethylaluminum Chloride (DEAC) [CAS: 96-10-6] is an organoaluminum compound widely utilized as a powerful reducing agent and co-catalyst. Its importance stems from its ability to significantly enhance polymerization processes, chemical synthesis efficiency, and reaction selectivity in various industries, including plastics, pharmaceuticals, and electronics.
Critical Importance in Modern Industry
The extensive use of DEAC arises from its ability to significantly boost chemical reactions, especially in polymer synthesis. For example, when combined with titanium compounds in Ziegler–Natta catalysis, DEAC accelerates polymerization reactions, enabling large-scale production of polyethylene and polypropylene. Its highly reactive nature makes it indispensable in catalytic processes, facilitating controlled polymer growth and improving the efficiency of catalytic systems. Moreover, DEAC is essential in chemical synthesis processes due to its capacity to act as a precise reagent, reducing unwanted by-products, thus maximizing yields and purity levels.
Despite these benefits, DEAC handling requires stringent safety measures, as it reacts violently upon contact with moisture or air. Proper storage and handling protocols must be strictly followed. Industrial plants utilizing DEAC must invest in training and safety equipment, ensuring staff safety and regulatory compliance.
| Industry Application | Significance |
|---|---|
| Polymerization | Increases reaction rates and selectivity |
| Pharmaceutical Synthesis | Enables precise, selective synthesis of drug intermediates |
| Specialty Chemicals | Provides critical catalytic action for specialty products |
Environmental and Regulatory Considerations
DEAC also demands compliance with environmental regulations due to potential hazards. Companies must navigate complex regulations to ensure safe, responsible use, often involving significant investments in regulatory compliance measures.
What Are the Key Chemical and Physical Properties of Diethylaluminum Chloride?
Understanding DEAC's chemical and physical properties is vital but often overlooked, leading to mishandling and potential hazards. Inadequate knowledge can compromise safety and process efficiency. A clear grasp of its properties resolves this.
Diethylaluminum Chloride [CAS: 96-10-6] is a colorless to pale-yellow liquid, highly reactive, sensitive to air and moisture. It exhibits pyrophoric characteristics, igniting spontaneously in air. Its chemical formula is (C₂H₅)₂AlCl, with key properties being high volatility, solubility in hydrocarbons, and strong reducing power.
Essential DEAC Properties
A comprehensive understanding of DEAC's chemical properties highlights its utility and the need for careful handling. As a pyrophoric substance, DEAC requires specialized storage under inert atmospheres (usually nitrogen or argon) to avoid spontaneous ignition upon contact with air. The compound has a boiling point around 127°C, which makes it volatile and relatively easy to handle under controlled conditions. Its high solubility in organic solvents like hexane and toluene ensures versatility across various chemical reactions.
| Property | Description |
|---|---|
| Chemical formula | (C₂H₅)₂AlCl |
| Appearance | Colorless to pale-yellow liquid |
| Reactivity | High (pyrophoric) |
| Boiling Point | Approx. 127°C |
| Solubility | Soluble in hydrocarbons |
Safety Implications
Given its properties, strict adherence to safe handling protocols is essential. Regular training and appropriate storage equipment are necessary for risk mitigation in industrial settings.
How Is Diethylaluminum Chloride Used in Ziegler–Natta Catalysis?
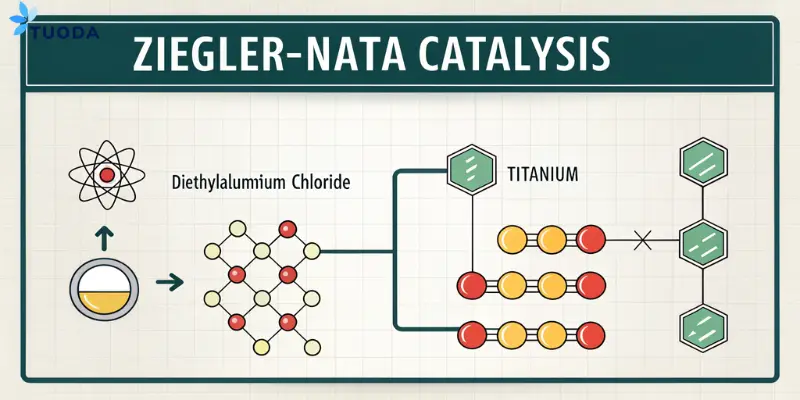
Companies often struggle to optimize polymerization processes, leading to increased costs and lower-quality polymers. Misuse or incorrect application of DEAC in catalysis can cause poor efficiency. Proper application addresses this issue effectively.
Diethylaluminum Chloride [CAS: 96-10-6] serves as an essential co-catalyst in Ziegler–Natta catalysis. It activates titanium-based catalysts, significantly enhancing polymerization reactions. DEAC increases catalytic activity and improves control over polymer characteristics, particularly in manufacturing polyethylene and polypropylene.
Role in Polymerization
In Ziegler–Natta polymerization, DEAC's role is to activate and sustain catalyst activity, enabling the production of high molecular weight polymers. Its use results in precise control over polymer microstructure, improving mechanical strength, flexibility, and durability. Industries benefit greatly by producing plastics with tailored properties suited to specific applications, thus reducing material wastage and enhancing product performance.
Advantages of DEAC in Polymerization
| Advantages | Explanation |
|---|---|
| Improved polymer quality | Consistent, high-quality polymers |
| Increased catalyst efficiency | Higher productivity and lower costs |
| Enhanced product customization | Precise control over polymer properties |
Why Is Diethylaluminum Chloride Valuable in Pharmaceutical Synthesis?
Pharmaceutical manufacturers face challenges ensuring purity and efficiency. Missteps during synthesis can produce impurities, risking regulatory rejection. DEAC ensures precise synthesis, mitigating these risks.
DEAC [CAS: 96-10-6] is valuable in pharmaceutical synthesis due to its powerful reducing capability and selectivity in reactions. It facilitates the production of complex pharmaceutical intermediates, ensuring high purity and minimizing undesired side reactions, thus enabling compliance with strict pharmaceutical regulations.
Impact on Drug Production
Pharmaceutical manufacturers increasingly rely on DEAC for synthesizing active pharmaceutical ingredients (APIs) and intermediates. It supports precise, efficient reactions required in complex drug synthesis. By reducing unwanted by-products and enabling reaction selectivity, DEAC helps manufacturers meet stringent quality and purity standards set by regulatory agencies like FDA or EMA.
| Pharmaceutical Use | Benefits |
|---|---|
| Drug intermediates synthesis | High purity, controlled reactions |
| Reducing agent | Minimizes impurities, regulatory compliance |
What Role Does Diethylaluminum Chloride Play in Fine and Specialty Chemicals?

Producing specialty chemicals demands precision, but manufacturers face risks of inconsistency and contamination. DEAC provides precise control, overcoming these issues.
In specialty chemical production, DEAC [CAS: 96-10-6] functions as a selective catalyst, enabling complex organic transformations with high efficiency. It enhances reaction rates and selectivity, ensuring high-quality, specialty products tailored for specific market needs such as fragrances, adhesives, and high-performance coatings.
Enhancing Specialty Chemicals Production
In fine chemical manufacturing, DEAC enables producers to create specialized products with greater efficiency and purity. Its catalytic properties offer flexibility in complex synthesis pathways, reducing waste and improving environmental compliance.
How Is Diethylaluminum Chloride Applied in the Electronics and Semiconductor Industries?

Semiconductor production demands extreme purity and precision. Errors in chemical processes cause significant financial losses. DEAC ensures accuracy and efficiency.
DEAC [CAS: 96-10-6] is utilized in electronics as a precursor and reagent in chemical vapor deposition (CVD) processes. It enables deposition of thin, high-purity aluminum-based films essential for semiconductor fabrication, improving electronic component performance and reliability.
What Are the Best Practices for Handling and Storing Diethylaluminum Chloride Safely?
Improper handling of DEAC poses significant safety risks. Without adequate safety protocols, accidents and environmental hazards occur. Implementing best practices resolves these dangers.
Safe handling practices include storing DEAC [CAS: 96-10-6] under inert atmospheres, using moisture-free environments, proper PPE, specialized containers, and rigorous employee safety training to prevent accidents and ensure compliance.
What Are the Current Market Trends and Global Demand for Diethylaluminum Chloride?

Unclear market trends complicate strategic procurement. Awareness of DEAC market developments guides efficient sourcing strategies.
The global demand for DEAC [CAS: 96-10-6] is growing, driven by increasing polymer production, pharmaceutical advancements, and electronics manufacturing. Asia-Pacific, notably China and India, dominates consumption due to expanding industrial activities.
Conclusion
DEAC [CAS: 96-10-6] is essential for polymer, pharmaceutical, specialty chemical, and electronics industries, requiring stringent handling to maximize its industrial benefits safely. Its versatility as a co-catalyst and reagent continues to drive innovation across multiple sectors, while proper understanding of its properties ensures both optimal performance and operational safety.
[1]:Explore this link to understand the diverse applications and properties of Diethylaluminum Chloride, enhancing your knowledge in industrial chemistry.
[2]:Learn about the polymerization process and the crucial role of Diethylaluminum Chloride in enhancing reaction rates and selectivity.
[3]:Discover essential safety measures for handling Diethylaluminum Chloride to ensure safe operations in industrial environments.









