
Chemical manufacturers struggle with inefficient catalytic processes that waste resources and increase costs. TEA offers a powerful solution that revolutionizes reaction efficiency while maintaining strict safety protocols.
Triethylaluminum (TEA, CAS 97-93-8) is an organometallic compound widely used as a co-catalyst in polymerization processes, particularly in Ziegler-Natta catalysis for polyolefin production. It also serves as an important reagent in organic synthesis and semiconductor manufacturing.
At TUODA, we've seen firsthand how the right catalyst can transform manufacturing efficiency. Let's explore why TEA has become indispensable in modern chemical processes and how it can benefit your operations.
What is Triethylaluminum (TEA) and Why is it Important?
Industries face increasing pressure to improve process efficiency while meeting stringent quality standards. TEA addresses these challenges by serving as a versatile reagent with exceptional reactivity and selectivity.
Triethylaluminum[^1] (Al(C₂H₅)₃, CAS 97-93-8) is an organometallic compound that functions as a powerful co-catalyst, alkylating agent, and reducing agent in various industrial applications. Its high reactivity with oxygen makes it valuable for oxygen scavenging in polymerization processes.
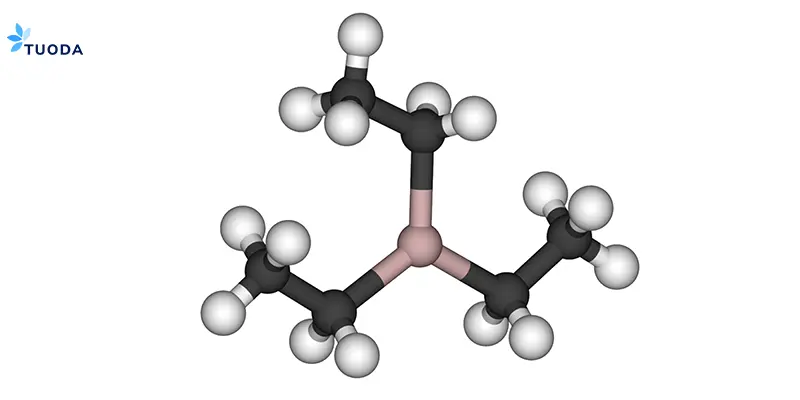
TEA's unique chemical structure enables it to:
- Form strong coordination complexes with transition metals
- Transfer ethyl groups to catalytic centers
- Scavenge oxygen, water, and other impurities from reaction systems
- Initiate polymerization reactions through precise activation mechanisms
The compound is a colorless liquid (bp: 194°C), typically supplied in hydrocarbon solvents at 10–25% for safer handling. Its pyrophoric nature requires specialized handling but contributes to its reactivity value.
What Are the Key Industrial Applications of Triethylaluminum?
TEA is indispensable in:
Polyolefin production[^2], semiconductor manufacturing[^3], fine chemical synthesis, and petrochemical processing.
Polymerization Catalyst Systems
TEA enables:
- Higher polymer yields
- Control over molecular weight
- Improved stereoselectivity (especially in polypropylene)
- Enhanced catalyst efficiency
Semiconductor Manufacturing
In CVD processes, 99.999% purity TEA ensures:
- Uniform thin films
- Minimal contamination
- Consistent electrical performance
Organic Synthesis Applications
| Reaction Type | TEA Function | Typical Products |
|---|---|---|
| Friedel-Crafts Alkylation | Lewis acid catalyst | Alkylated aromatics |
| Reduction Reactions | Hydride source | Reduced compounds |
| C–C Bond Formation | Organometallic reagent | Complex molecules |
| Ring-Opening Polymerization | Initiator | Specialty polymers |
Petrochemical Processing
- Catalyst Activation
- Oligomerization of light olefins
- Impurity Removal (e.g., sulfur scavenging)
How is Triethylaluminum Produced Commercially?
Produced via direct synthesis from aluminum, hydrogen, and ethylene. Alternative methods include transalkylation.

Direct Synthesis
Raw Materials
- High-purity aluminum
- Ethylene (<5 ppm moisture)
- Hydrogen (ultra-pure)
Reaction Mechanism
- Aluminum activated by halides or TEA
- Hydrogen forms Al–H intermediates
- Ethylene inserts into Al–H
- Stepwise formation of TEA
| Parameter | Typical Range | Impact |
|---|---|---|
| Temp | 120–180°C | Affects rate & side reactions |
| Pressure | 20–30 bar | Ethylene solubility |
| Catalyst concentration | 0.1–1.0 mol% | Reaction rate |
| Ethylene ratio | 0.8–1.2 | Conversion efficiency |
Purification
- Fractional distillation
- Crystallization (ultra-pure grades)
- Filtration (final stage)
What Safety Protocols Are Essential When Handling TEA?
TEA is pyrophoric — ignites in air. Requires inert systems, PPE, and dry chemical fire suppression.

Safety Framework
- Storage
- 15–25°C, inert blanket, sensors
- Transfer
- Closed-loop systems, nitrogen purge
- Personnel
- Fire-resistant gear, training, SCBA
- Emergency
- Shutdowns, local responder coordination
What is the Role of TEA in Ziegler-Natta Catalysis?
TEA reduces cost and increases control in Ziegler-Natta polymerization.
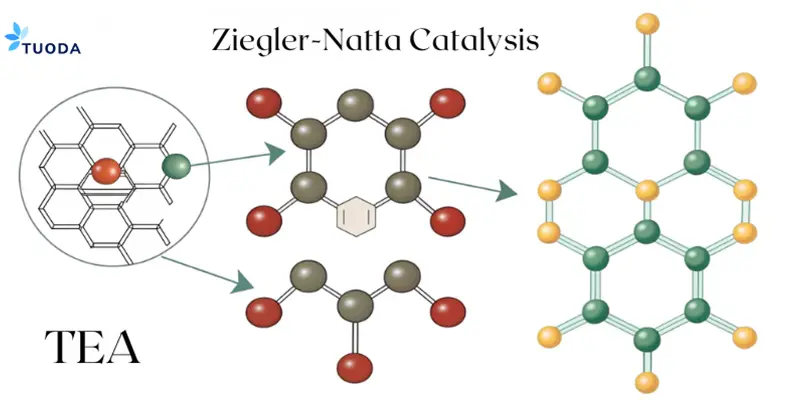
Mechanisms
| Function | Process | Catalysis Impact |
|---|---|---|
| Alkylation | Ti–Cl + TEA → Ti–Et | Creates active sites |
| Reduction | Ti⁴⁺ → Ti³⁺ by TEA | Enhances activity |
| Scavenging | H₂O + TEA → TEA–OH + C₂H₆ | Removes poisons |
| Chain Transfer | Ti-polymer + TEA → Ti–Et + polymer–TEA | MWD control |
Concentration Effects
- Al/Ti ratios: 50:1 – 300:1
- Optimal TEA improves quality and lowers cost
How Does TEA Compare to Other Aluminum Alkyls?
| Alkyl | Structure | CAS | Advantages | Applications |
|---|---|---|---|---|
| TEA | Al(C₂H₅)₃ | 97-93-8 | Reactive, selective, cost-effective | HDPE, LLDPE |
| TIBA | Al(i-C₄H₉)₃ | 100-99-2 | Stereoregularity control | Polypropylene |
| DEAC | Al(C₂H₅)₂Cl | 96-10-6 | High acidity, MWD control | Elastomers |
| MAO | [Al(CH₃)O]ₙ | 146905-79-5 | Metallocene activation | Specialty polymers |
Selection Criteria
- Polymer type
- Catalyst compatibility
- Cost and safety considerations
What Quality Specifications Are Critical for TEA in Industrial Applications?
| Parameter | Spec | Method | Performance Impact |
|---|---|---|---|
| Aluminum Content | 22.0–23.0 wt% | Titration | Determines activity |
| Hydrolyzable Ethyl | >95% theoretical | Gas analysis | Reactivity check |
| Chloride Content | <50 ppm | IC | Catalyst poison indicator |
| Transition Metal Impurities | <5 ppm | ICP-MS | Affects side reactions |
| Free Ethane | <0.5 wt% | GC | Stability indicator |
| Appearance | Clear liquid | Visual | General quality |
Application-Specific
- Semiconductor: 99.999% purity, no Fe/Cu/Cr
- Polyolefins: Ultra-low metals, clean ethyl groups
How Can TEA Optimization Improve Manufacturing Economics?
TEA can improve:
- Catalyst productivity: +15–30%
- Off-spec rate: –40%+
- Overall cost: Major reductions
Optimization Strategies
- Staged addition
- Temperature-dependent dosing
- Scavenger balancing
- Dosing systems
Case Study: Polyethylene Production
| Parameter | Before | After | Change |
|---|---|---|---|
| TEA Usage | 2.8 kg/ton | 2.1 kg/ton | ↓ 25% |
| Catalyst Productivity | 8,500 → 10,200 | ↑ 20% | |
| Off-spec Rate | 3.2% → 1.8% | ↓ 44% | |
| Annual Cost Saving | - | - | $1.2 million |
Conclusion
Triethylaluminum (CAS 97-93-8) plays a vital role in polymerization, fine chemicals, and semiconductor industries. Its proper handling and optimization offer immense opportunities for cost savings and efficiency improvements.
[^1]: Explore this link to understand the significance of Triethylaluminum in various industries and its unique properties.
[^2]: Discover how Triethylaluminum enhances polyolefin production and its role in catalyst systems for better yields.
[^3]: Learn about the critical functions of Triethylaluminum in semiconductor processes and its impact on device performance.









